
We encourage researchers, visual designers, and educators to upload and share their works to demonstrate the creative potential of ChemBlender. Through collective contribution, we hope to promote broader participation in its application and development, and to advance the progress of scientific visualization.
E-mail: lihaodong@ustc.edu.cn
Ju Huang, et al. ACS Nano, 2026, 20(1): 565-574.
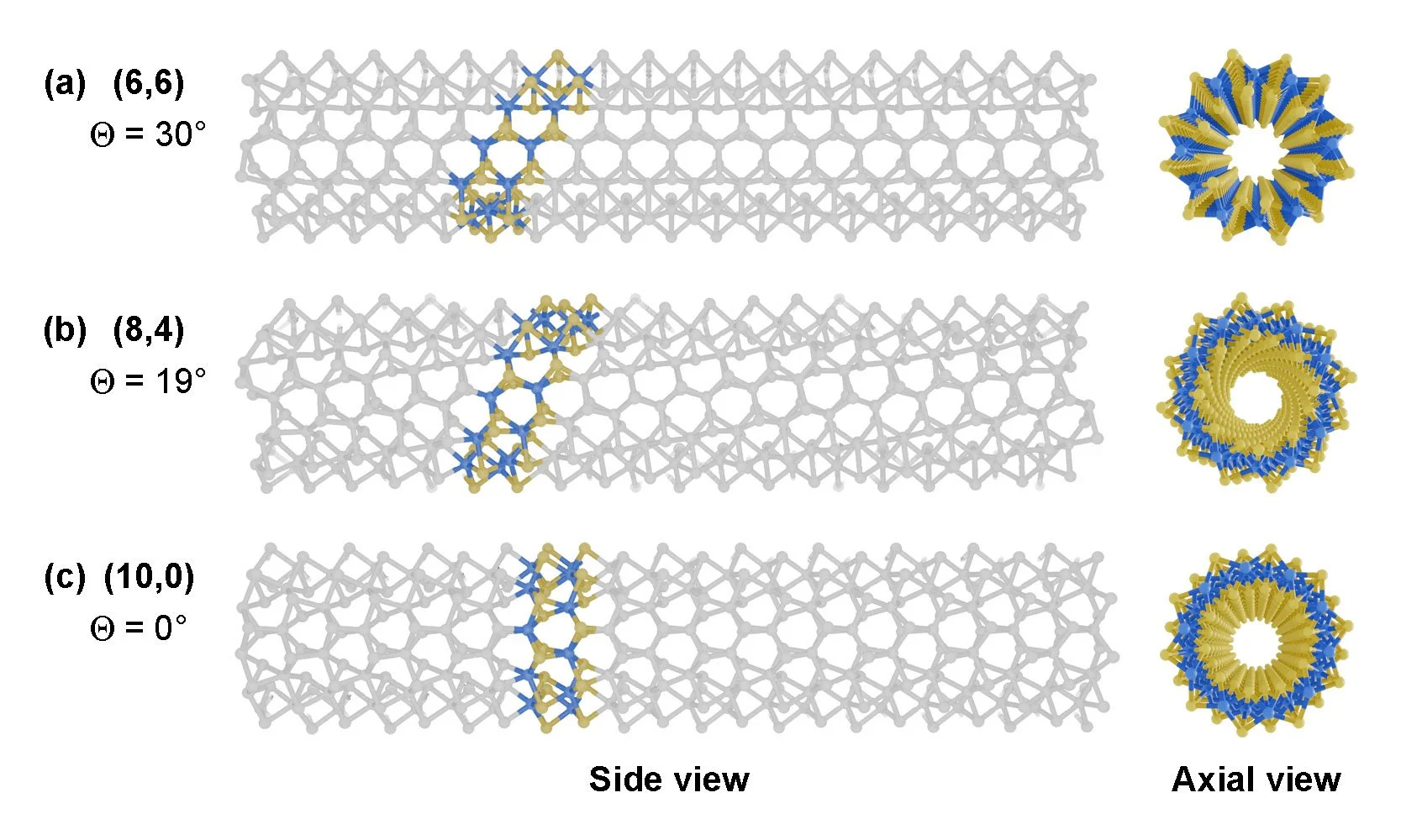
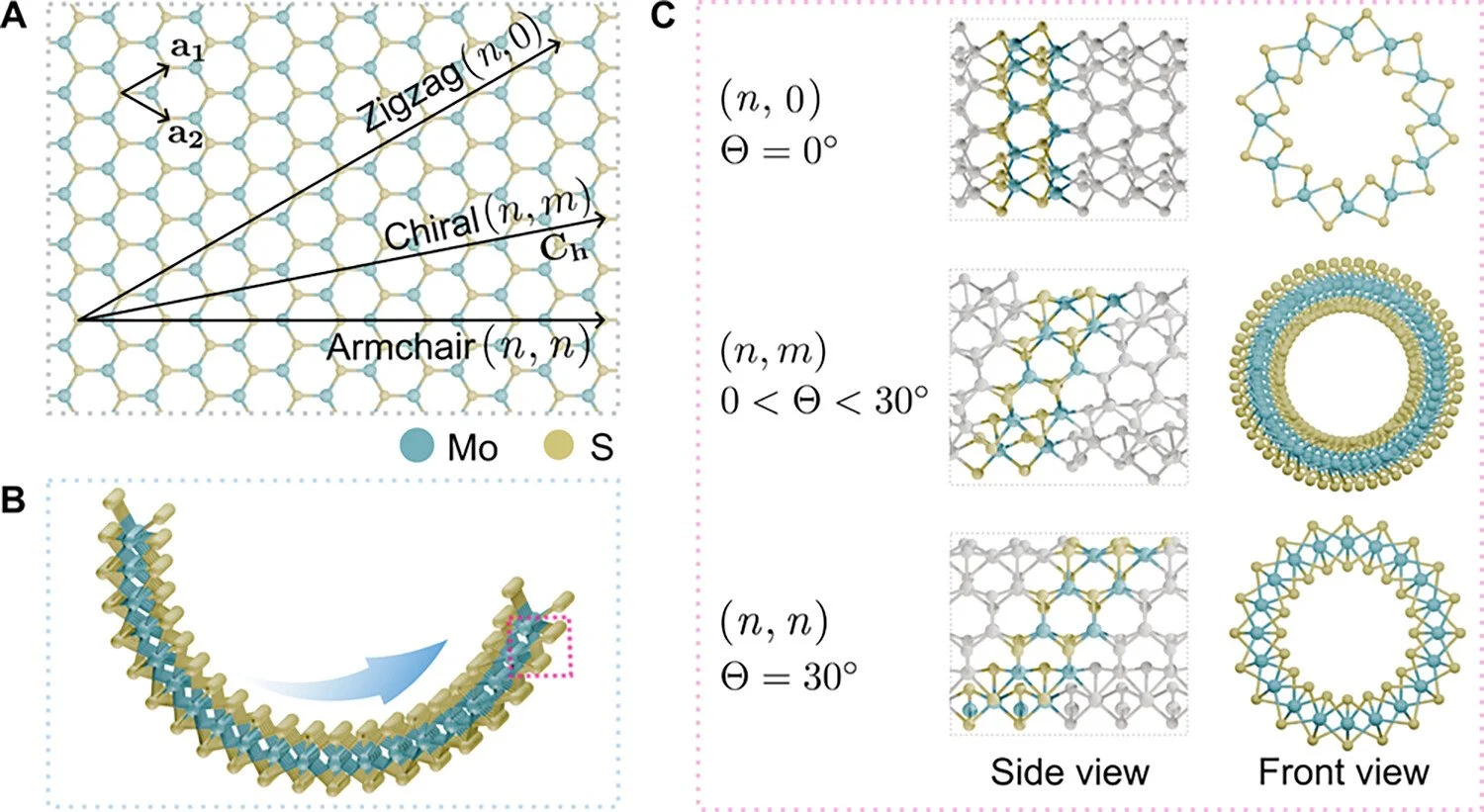
Similar to carbon nanotubes, MoS₂ nanotubes can also adopt zig-zag, arm-chair, and chiral structures, depending on the rolling direction of the MoS₂ layers, which significantly influence their electronic and mechanical properties.
As shown in the figure, the side views and axial views of different MoS₂ nanotubes were modeled and rendered using the Chem Nodes preset node groups. The chiral indices of (a)–(c) are (6,6), (8,4), and (10,0), respectively.
To enhance the clarity of the side view, a segmented alpha value was applied during rendering to gradually fade out the rear structure.
Figure 1. MoS2 nanotubes with intrinsic structural chiralities. (A) Top view of a MoS2 monolayer. The structural parameters of nanotubes are illustrated, including the two-dimensional (2D) basis vectors a1 and a2, and the chiral vector Ch = na1 + ma2 ≡ (n, m), with n ≥ m ≥ 0. (B) Side view of rolling and zipping a MoS2 nanoribbon to form a nanotube following a chiral vector illustrated in (A). (C) Side and front views of zigzag, chiral, and armchair MoS2 nanotubes, which correspond to chiral indices (n, 0), (n, m), and (n, n), respectively. The chiral angle Θ is defined as the angle between the chiral vector Ch and the a1 direction. Zigzag and armchair nanotubes have chiral angles of 0 and 30°, respectively, while chiral nanotubes have a chiral angle between these values. The expressions of additional parameters, such as the one-dimensional translational vector T of nanotube corresponding to Ch.
Shujian Li, et al. Journal of Colloid Interface Science, 2026, 708: 139865.
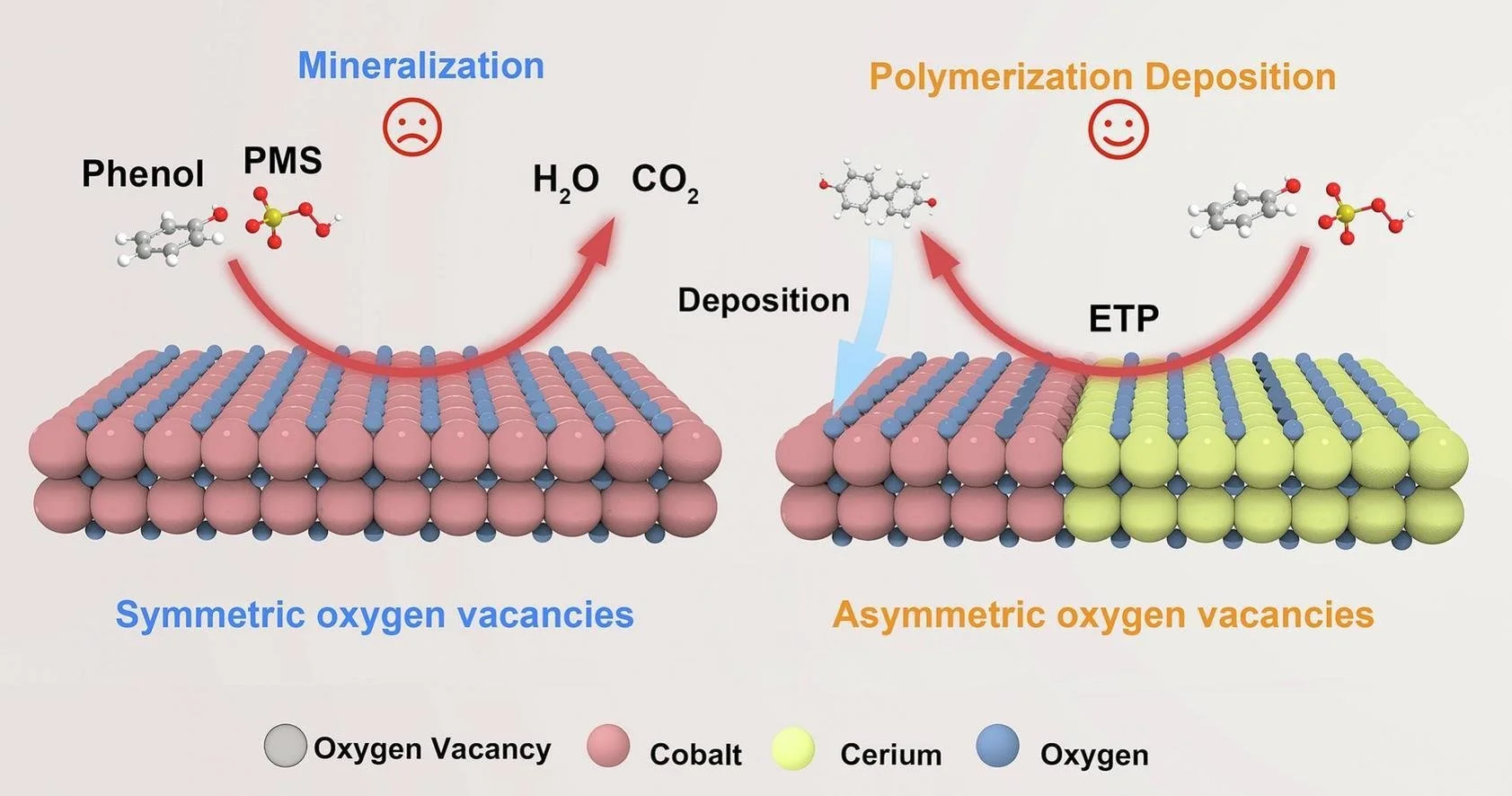
Polymerization–deposition (PD) pathway offers a promising route for low-carbon wastewater treatment, yet their efficiency under ultra-low oxidant dosage remains challenging. Herein, interfacial asymmetric oxygen vacancies (As-Ov) were constructed in hollow Co3O4/CeO2 composites (H-Co30Ce-Ov) using glucose-derived hydrothermal carbon microspheres as sacrificial templates. The interfacial As-Ov drives an electron-transfer pathway (ETP) for phenol (PN) removal via peroxymonosulfate (PMS) activation to generate phenoxy radicals. These radicals subsequently underwent coupling reactions to form oligomeric products, resulting in effective COD removal via a PD-dominated process under ultra-low PMS dosage. Mechanistic investigations, including in situ Raman and electrochemical analyses, confirms the critical role of the coordination environment of As-Ov in controlling PMS activation behavior and reaction kinetic. Moreover, the H-Co30Ce-Ov exhibited excellent catalytic activity toward electron-rich pollutants, high PMS utilization efficiency and good stability across different water matrices.
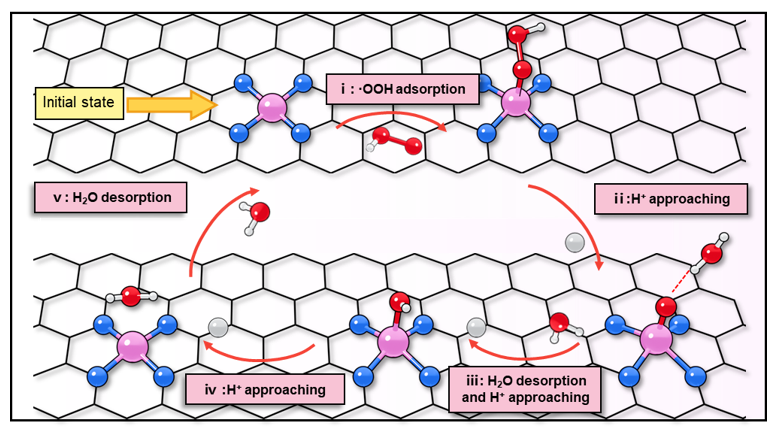
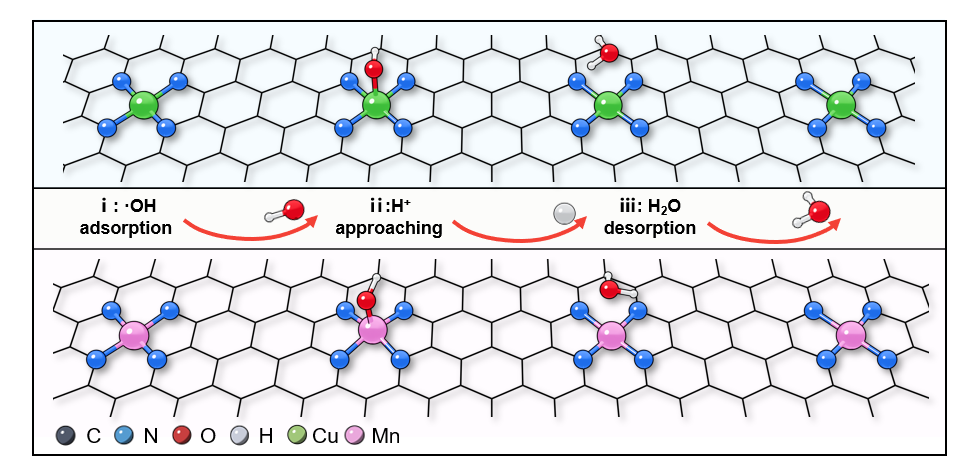
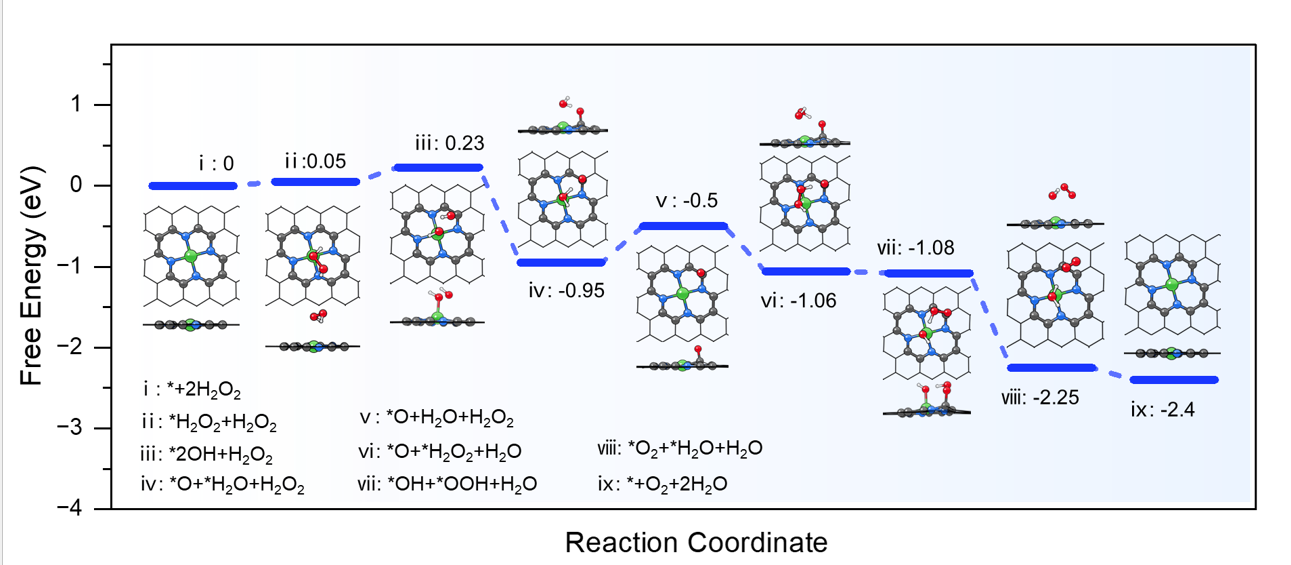
Fucheng Gao, et al. Science Bulletin, 2025, 70(17): 2730-2734. (Supporting Information)
https://doi.org/10.1016/j.scib.2025.07.004 Download: Download Acrobat PDF file (9MB)